SELECTED IMPORTANT SAFETY INFORMATION: JIVI is contraindicated in patients who have a history of hypersensitivity reactions to the active substance, polyethylene glycol (PEG), mouse or hamster proteins, or other constituents of the product. CONTINUE READING BELOW
This site is intended for US residents.
SELECTED IMPORTANT SAFETY INFORMATION: JIVI is contraindicated in patients who have a history of hypersensitivity reactions to the active substance, polyethylene glycol (PEG), mouse or hamster proteins, or other constituents of the product. CONTINUE READING BELOW
SELECTED IMPORTANT SAFETY INFORMATION: JIVI is contraindicated in patients who have a history of hypersensitivity reactions to the active substance, polyethylene glycol (PEG), mouse or hamster proteins, or other constituents of the product. CONTINUE READING BELOW
JIVI® VS ELOCTATE® PK CROSSOVER STUDY
First read about the PK parameters of Jivi® from the PROTECT VIII trial, which will be helpful to understanding the data below from the crossover study examining the PK characteristics of Jivi® and Eloctate®.
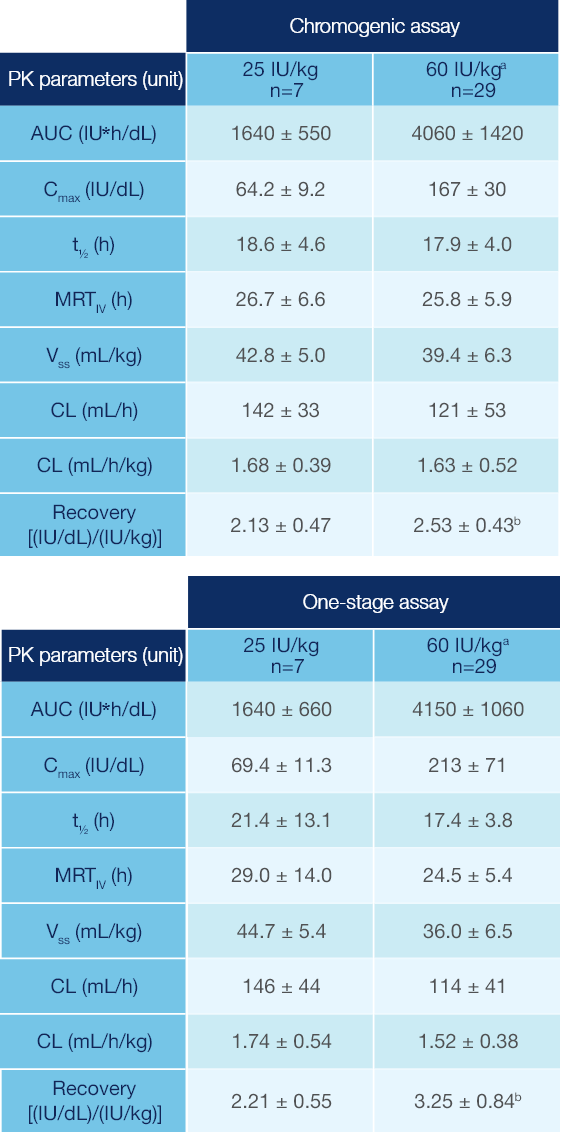
PK Parameters of Jivi® in Patients ≥12 Years of Age1
PK parameters of Jivi® in the PROTECT VIII trial (arithmetic mean ± SD)1
Measured following a single dose (25 IU/kg and 60 IU/kg)1
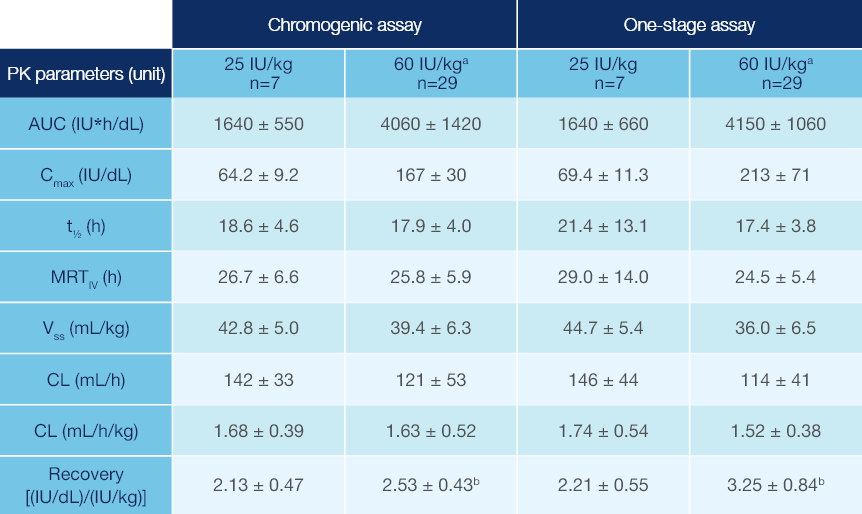
Combined data from Phase 1 and Phase 2/3 studies.
Recovery value could not be calculated for one subject.
PK: pharmacokinetic; SD: standard deviation; AUC: area under the curve; Cmax: maximum drug concentration in plasma after single dose;
t1/2: terminal half-life; MRTIV: mean residence time after an IV administration; VSS: apparent volume distribution at steady-state; CL: clearance.
Combined data from Phase 1 and Phase 2/3 studies.
Recovery value could not be calculated for one subject.
PK: pharmacokinetic; SD: standard deviation; AUC: area under the curve; Cmax: maximum drug concentration in plasma after single dose; t1/2: terminal half-life; MRTIV: mean residence time after an IV administration; VSS: apparent volume distribution at steady-state; CL: clearance.
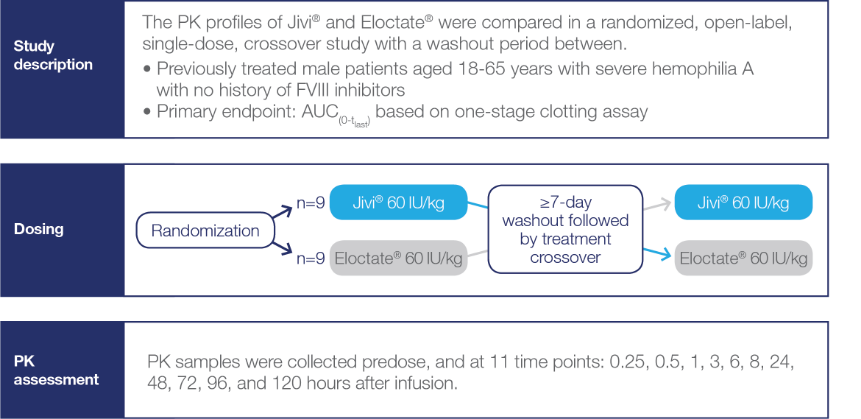
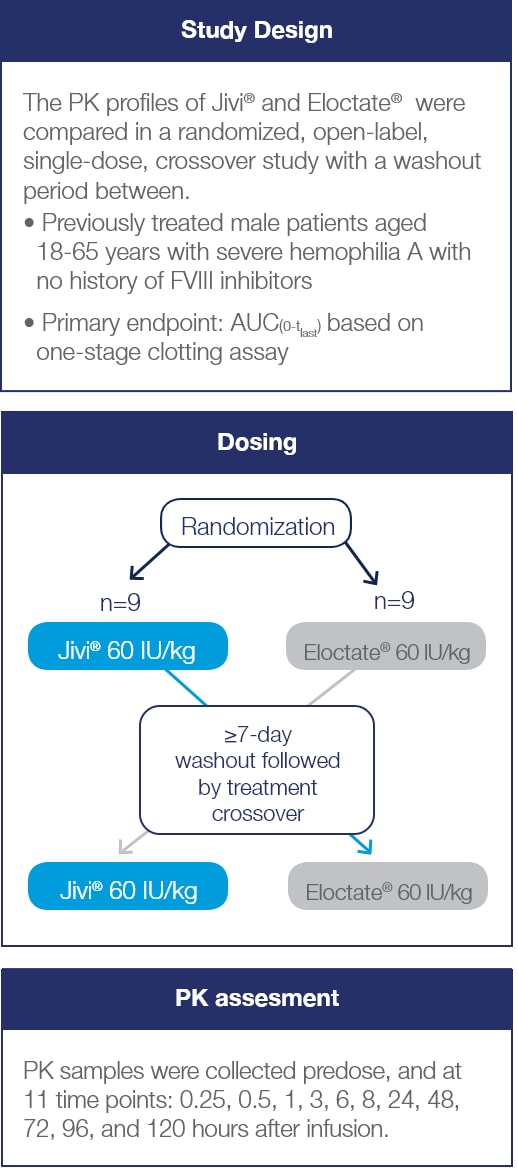
PK Crossover Study Design
Crossover study examining PK characteristics of Jivi® and Eloctate® (N=18)2*
AUC of Jivi® vs Eloctate®
Jivi® AUC compared to Eloctate® in 18 patients
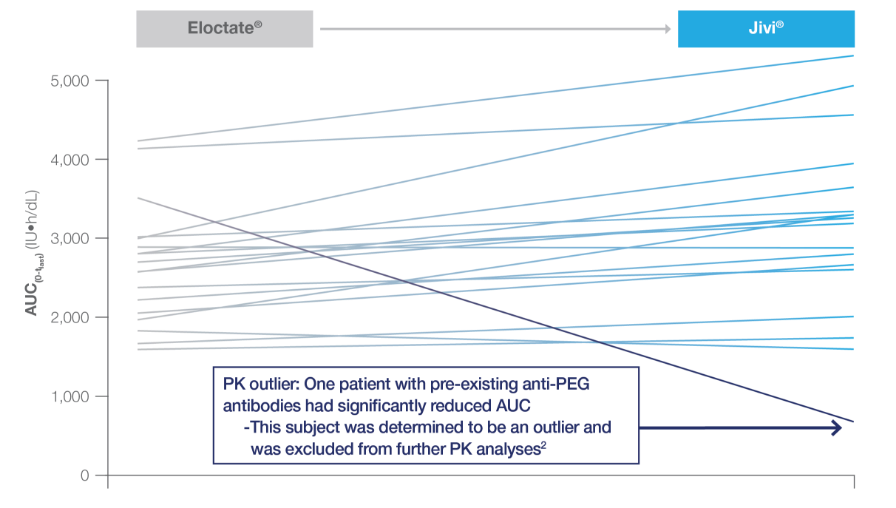
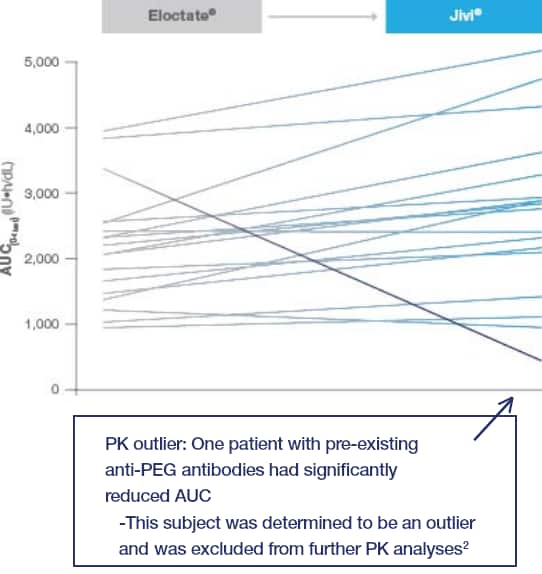
The pre-specified criteria for non-inferiority of Jivi® vs Eloctate® were met
Superiority criteria of the Jivi®: Eloctate® ratio were not met (95% CI 0.84–1.46; P=0.46)
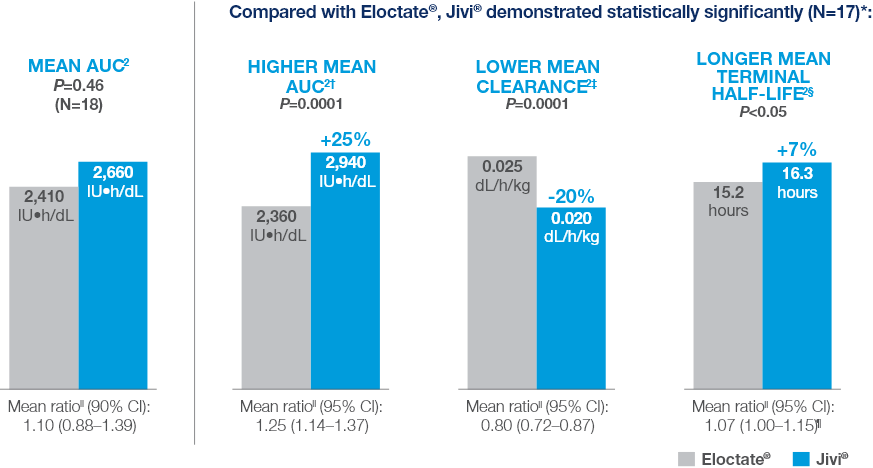
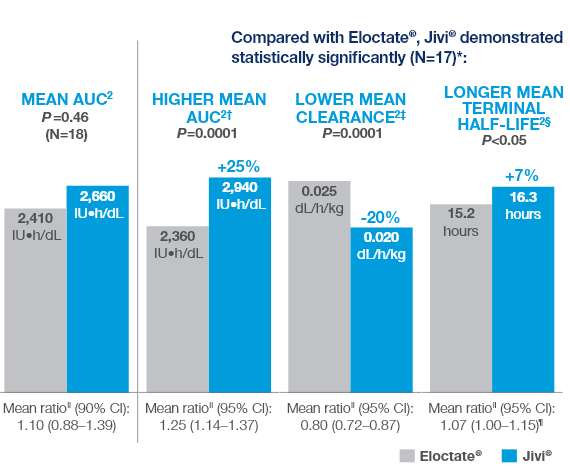
Comparative PK Results
Comparative PK Results for Jivi® vs Eloctate®2
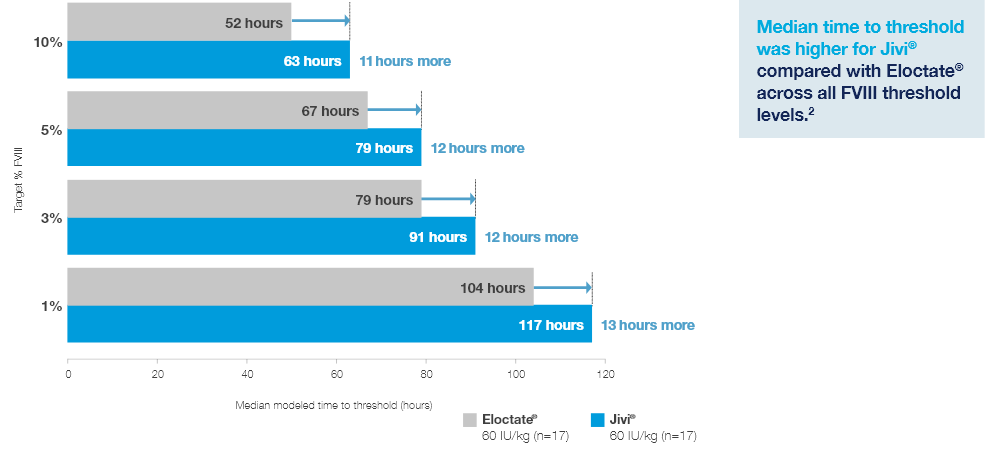
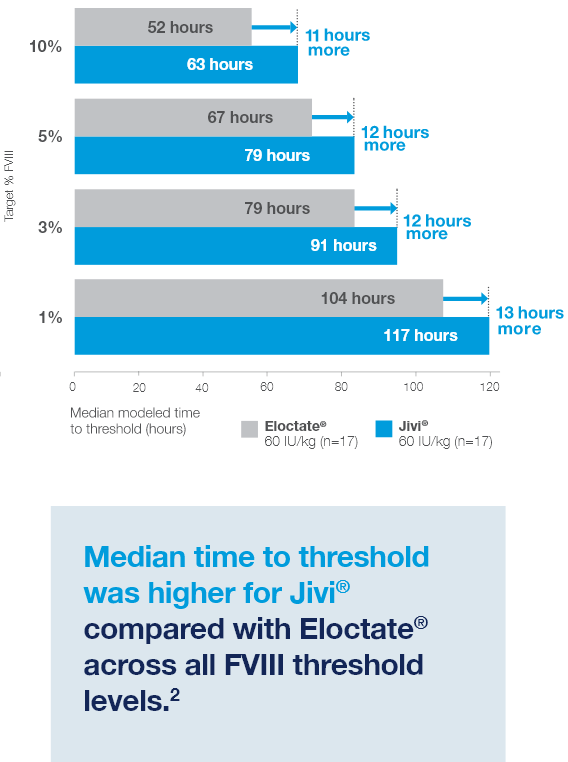
Time to Target Threshold Levels
Median time to target FVIII threshold levels with Jivi® vs Eloctate®2
Estimated from a population PK model (excluding outlier, N=17)2*
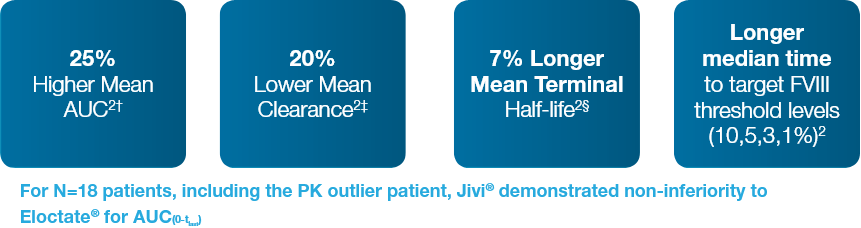
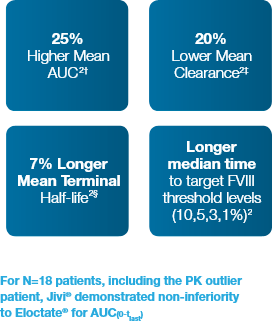
Summary of Comparative Results (Key Findings)
Compared with Eloctate®, Jivi® demonstrated statistically significantly (N=17)*:
INDICATION
JIVI® is a recombinant DNA-derived, Factor VIII concentrate indicated for use in previously treated adults and pediatric patients 7 years of age and older with hemophilia A (congenital Factor VIII deficiency) for:
On-demand treatment and control of bleeding episodes.
Perioperative management of bleeding.
Routine prophylaxis to reduce the frequency of bleeding episodes.
Limitations of use
JIVI is not indicated for use in:
Children <7 years of age due to a greater risk for hypersensitivity reactions and/or loss of efficacy.
Previously untreated patients (PUPs).
Treatment of von Willebrand disease.
IMPORTANT SAFETY INFORMATION
JIVI is contraindicated in patients who have a history of hypersensitivity reactions to the active substance, polyethylene glycol (PEG), mouse or hamster proteins, or other constituents of the product.
Hypersensitivity reactions, including severe allergic reactions, have occurred with JIVI. Monitor patients for hypersensitivity symptoms. Early signs of hypersensitivity reactions, which can progress to anaphylaxis, may include chest or throat tightness, dizziness, mild hypotension and nausea. If hypersensitivity reactions occur, immediately discontinue administration and initiate appropriate treatment.
JIVI may contain trace amounts of mouse and hamster proteins. Patients treated with this product may develop hypersensitivity to these non-human mammalian proteins.
Hypersensitivity reactions may also be related to antibodies against polyethylene glycol (PEG).
Neutralizing antibody (inhibitor) formation has occurred following administration of JIVI. Carefully monitor patients for development of Factor VIII inhibitors, using appropriate clinical observations and laboratory tests. If expected plasma Factor VIII activity levels are not attained or if bleeding is not controlled as expected with administered dose, suspect the presence of an inhibitor (neutralizing antibody).
An immune response associated with IgM anti-PEG antibodies, manifested as symptoms of acute hypersensitivity and/or loss of drug effect, has occurred with JIVI administration. In the clinical trials, the IgM anti-PEG antibodies disappeared within 4-6 weeks. No immunoglobulin class switching from IgM to IgG has been observed.
A low post-infusion Factor VIII level, in absence of detectable Factor VIII inhibitors, may be due to loss of treatment effect related to high titers of anti-PEG IgM antibodies. In these cases, discontinue JIVI and switch patients to a different anti-hemophilic product.
A reduced recovery of Factor VIII after start of JIVI treatment may be due to transient low titers of anti-PEG IgM antibodies. In these cases, increase the dose of JIVI until recovery of Factor VIII returns to expected levels.
The most common (incidence ≥5%) adverse reactions in clinical trials in previously treated patients (PTPs) ≥7 years of age were headache, fever, cough, and abdominal pain.
For additional important risk and use information, please see the full Prescribing Information.
You are encouraged to report side effects or quality complaints of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088. For Bayer products, you can report these directly to Bayer by clicking here.
INDICATION
JIVI® is a recombinant DNA-derived, Factor VIII concentrate indicated for use in previously treated adults and pediatric patients 7 years of age and older with hemophilia A (congenital Factor VIII deficiency) for:
On-demand treatment and control of bleeding episodes.
Perioperative management of bleeding.
Routine prophylaxis to reduce the frequency of bleeding episodes.
Limitations of use
JIVI is not indicated for use in:
Children <7 years of age due to a greater risk for hypersensitivity reactions and/or loss of efficacy.
Previously untreated patients (PUPs).
Treatment of von Willebrand disease.
IMPORTANT SAFETY INFORMATION
JIVI is contraindicated in patients who have a history of hypersensitivity reactions to the active substance, polyethylene glycol (PEG), mouse or hamster proteins, or other constituents of the product.
Hypersensitivity reactions, including severe allergic reactions, have occurred with JIVI. Monitor patients for hypersensitivity symptoms. Early signs of hypersensitivity reactions, which can progress to anaphylaxis, may include chest or throat tightness, dizziness, mild hypotension and nausea. If hypersensitivity reactions occur, immediately discontinue administration and initiate appropriate treatment.
JIVI may contain trace amounts of mouse and hamster proteins. Patients treated with this product may develop hypersensitivity to these non-human mammalian proteins.
Hypersensitivity reactions may also be related to antibodies against polyethylene glycol (PEG).
Neutralizing antibody (inhibitor) formation has occurred following administration of JIVI. Carefully monitor patients for development of Factor VIII inhibitors, using appropriate clinical observations and laboratory tests. If expected plasma Factor VIII activity levels are not attained or if bleeding is not controlled as expected with administered dose, suspect the presence of an inhibitor (neutralizing antibody).
An immune response associated with IgM anti-PEG antibodies, manifested as symptoms of acute hypersensitivity and/or loss of drug effect, has occurred with JIVI administration. In the clinical trials, the IgM anti-PEG antibodies disappeared within 4-6 weeks. No immunoglobulin class switching from IgM to IgG has been observed.
A low post-infusion Factor VIII level, in absence of detectable Factor VIII inhibitors, may be due to loss of treatment effect related to high titers of anti-PEG IgM antibodies. In these cases, discontinue JIVI and switch patients to a different anti-hemophilic product.
A reduced recovery of Factor VIII after start of JIVI treatment may be due to transient low titers of anti-PEG IgM antibodies. In these cases, increase the dose of JIVI until recovery of Factor VIII returns to expected levels.
The most common (incidence ≥5%) adverse reactions in clinical trials in previously treated patients (PTPs) ≥7 years of age were headache, fever, cough, and abdominal pain.
For additional important risk and use information, please see the full Prescribing Information.
You are encouraged to report side effects or quality complaints of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088. For Bayer products, you can report these directly to Bayer by clicking here.
References: 1. Jivi® Prescribing Information. Whippany, NJ: Bayer LLC; May 2025. 2. Shah A, Solms A, Wiegmann S, et al. Direct comparison of two extended-half-life recombinant FVIII products: a randomized, crossover pharmacokinetic study in patients with severe hemophilia A. Ann Hematol. 2019;1-10. https://link.springer.com/article/10.1007%2Fs00277-019-03747-2. Published June 24, 2019. Accessed June 25, 2019. 3. Anderson PL. The ABCs of pharmacokinetics. http://www.thebody.com/content/art875.html. Accessed April 2018. 4. Dhillon S, Gill K. Basic pharmacokinetics. In: Dhillon S, Kostrzewski A, eds. Clinical Pharmacokinetics. London, UK: Pharmaceutical Press; 2006. 5. Ratain MJ, Plunkett WK Jr. Principles of pharmacokinetics. In: Kufe DW, Pollock RE, Weichselbaum RR, et al, eds. Holland-Frei Cancer Medicine. 6th ed. Hamilton, Ontario: BC Decker Inc; 2003. 6. Jivi vs Eloctate PK Confidence Interval Data Table from CSR. Bayer; 2019.






